Join us for a virtual Symposium on Open Guidelines for RDTs
What if the next time you opened any rapid diagnostic test (RDT) from any manufacturer for any condition, you can scan it with a WHO-approved and freely available smartphone app that would instantly recognize the RDT, authenticate it, support step-by-step instructions, read it with optimal sensitivity, interpret results based on the patient’s symptoms, medical record, local disease epidemiology, and issue curated recommendations for care, all in 15 minutes or less?
What if the next time a test was degraded due to poor storage or heat, or was expired, or counterfeit, you are immediately informed and know to use another test?
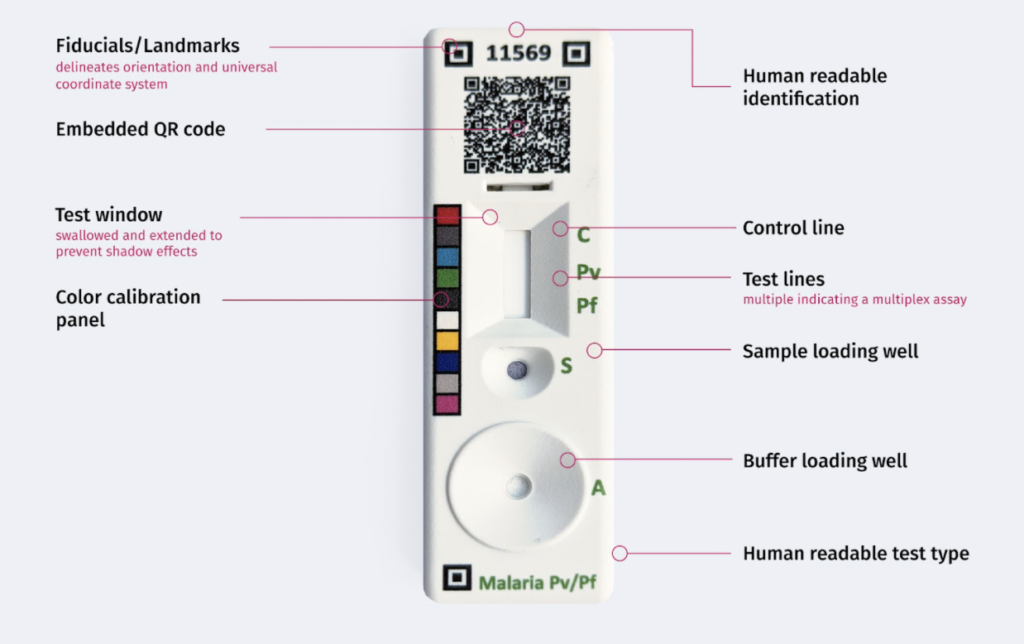
Over the past three years we have been supported by the Bill and Melinda Gates Foundation to address these questions. In partnership with the RDT manufacturing and implementation community we have defined a framework and conducted exploratory research and implementations towards open guidelines for precision healthcare. We are excited to discuss and present the community’s work during a virtual symposium on the future of Open Guidelines for RDTs.
During the symposium we will share and discuss advances in RDT, smartphone, and healthcare system integration. We will present results from our work developing and piloting experimental RDTs for Malaria and Covid-19, created in partnership with the manufacturing and implementation community based on a set of Open Guidelines. The virtual event will take place on March 10th and 11th, 2022 from 9am-1pm EST (GMT -5).
We have additional background on our RDT work available on the Ona Blog. If you would like to join us and attend the event, please email research@ona.io.

